Abstract
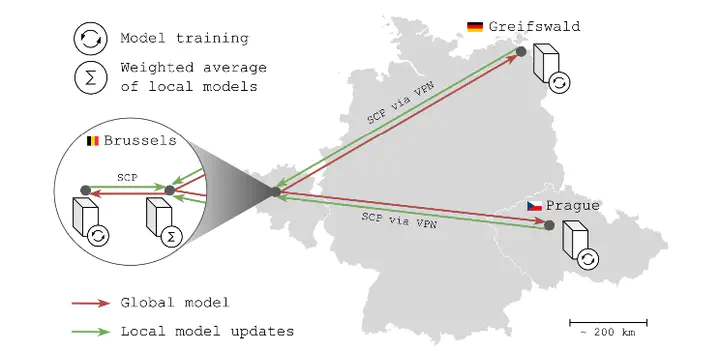
BACKGROUND: Federated learning (FL) has the potential to boost deep learning in neuroimaging but is rarely deployed in real-world scenarios, where its true potential lies. We propose FLightcase, a new FL toolbox tailored for brain research, and evaluate it on a real-world FL network to predict the cognitive status in patients with multiple sclerosis (MS) from brain magnetic resonance imaging (MRI). METHODS: We first trained a DenseNet neural network to predict age from T1-weighted brain MRI on three open-source datasets: IXI (586 images), SALD (491 images), and CamCAN (653 images). These were distributed across the three centres in our FL network: Brussels (BE), Greifswald (DE), and Prague (CZ). We benchmarked this federated model with a centralised version. The best-performing brain age model was then fine-tuned to predict performance on the symbol digit modalities test (SDMT) of patients with MS (Brussels: 96 images, Greifswald: 756 images, Prague: 2,424 images). Shallow transfer learning (TL) was compared with deep transfer learning, in which weights were updated either in the last layer or across the entire network, respectively. RESULTS: Federated training outperformed centralised training, predicting age with a mean absolute error (MAE) of 6.08 versus 7.02. Federated training yielded Pearson correlations (all p textless .001) between true and predicted age of0.88 (IXI, Brussels), 0.91 (SALD, Greifswald), and 0.93 (CamCAN, Prague). Fine-tuning of the centralised model to SDMT was most successful with a deep TL paradigm (MAE = 9.19) compared to shallow TL (MAE = 11.05). Across Brussels, Greifswald, and Prague, deep TL predicted SDMT with MAEs of 10.71, 9.67, and 8.98, respectively, and yielded Pearson correlations between true and predicted SDMT of.25 (p = 0.282), 0.40 (p textless 0.001), and 0.50 (p textless 0.001). CONCLUSION: Real-world federated learning using FLightcase is feasible for neuroimaging research in MS, enabling access to large MS imaging databases without sharing data. The federated SDMT-decoding model is promising and could be improved in the future by adopting FL algorithms that address the non-IID data issue and consider other imaging modalities. We hope our detailed real-world experiments and open-source distribution of FLightcase will prompt researchers to move beyond simulated FL environments.